So now we’re going to think about heat. And we’re going to think about all the little devices that we use. Microprocessors in our phones. About industrial-scale processes. Steel processing, oil refining. About transport, so planes, trains, and automobiles. And the heat that’s generated in those processes. You’ve all had a hot phone, I’m sure.

Machines generate waste heat when they do work for us. And this year, seven billion of us will use twenty-five trillion kilowatt hours of electricity. An awful lot of that will end up as waste heat. So, we treat waste heat as a problem. We see it as a challenge to design how we can manage it. We don’t think of it as a resource. If we thought of it as a resource, that would be results we are just throwing away.

But it’s worse than that. We’re not just wasting heat, we’re using even extra energy trying to manage that heat. So extra systems on aircraft, for example, to keep the system cool. At the other end of our thermal spectrum, refrigeration and air conditioning is now using something like 15% of our global electricity production.

So here we are in the 21st century. Wouldn’t it be great if we could do something better? If we could use that heat to extract electricity. Or if we could use electricity more efficiently. Cool systems, systems we could just embedded into everyday objects, that would allow us to have electricity back to power systems.
So, why aren’t we doing that? Well, there are a few problems. But really they’re both two sides of the same coin. How can we use heat to extract electricity? How can we use electricity to extract cooling? They’re about material systems that have transitions in them. And we could start to address some of those problems if we think a bit more cleverly about how we do materials engineering.
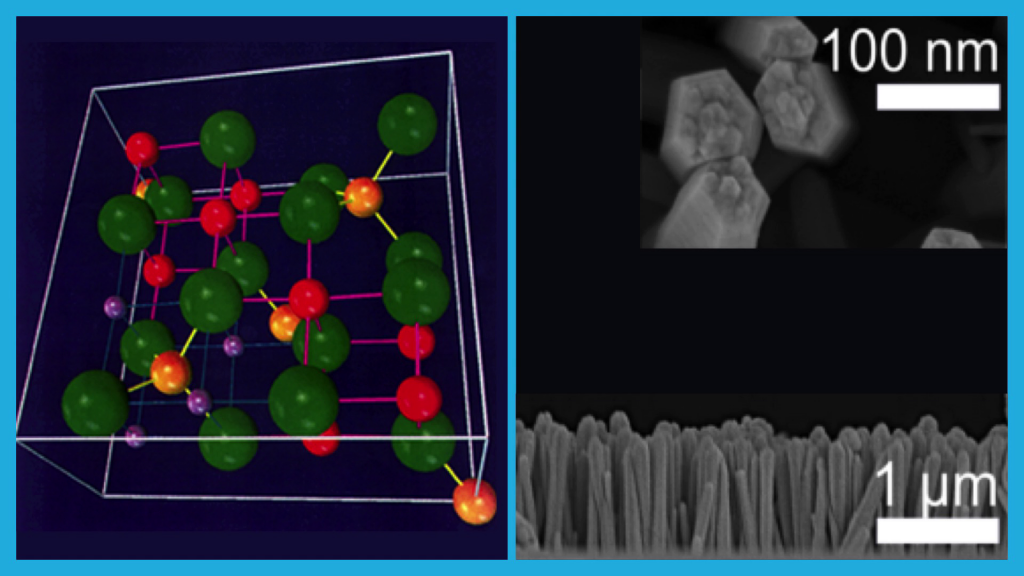
So one of the key challenges is one of chemistry. How do we get the atomic structure of the materials we’re using right so that they do effectively and efficiently couple between thermal and electrical energy? The second problem is one of structure. How do you get materials that can pump effectively electricity or heat in and out of those systems? We’re going to need new materials combined at the nanoscale to be able to do this.
So when we talk about chemistry and nanostructure, we only have to look around us to see lots of examples. Nature does this naturally. Beautiful, interconnected systems. And we can start to look to biology to think about ways to both design and to start to grow new nanoscale nanocomposite systems.
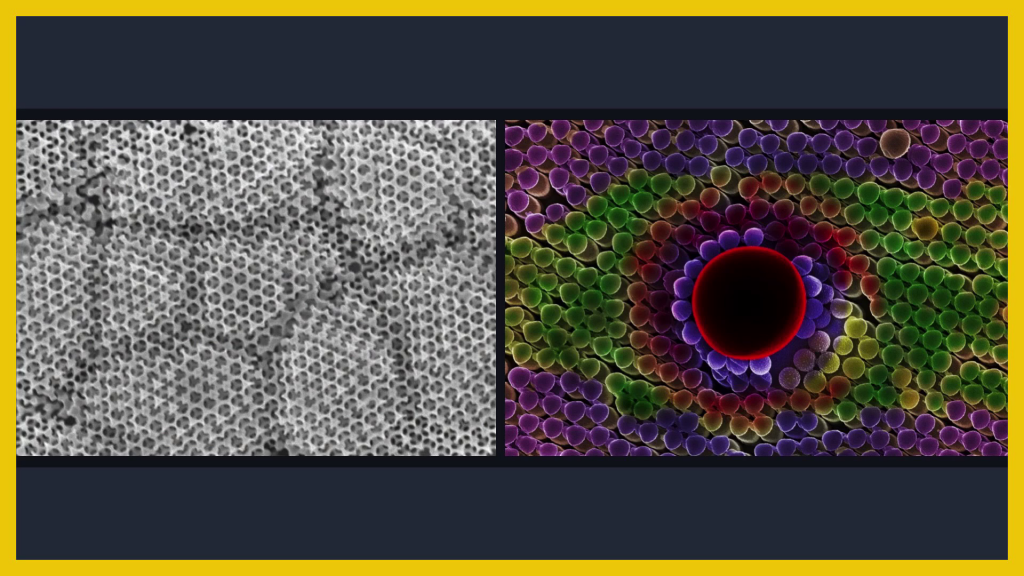
You might have multiple layers of materials, just atomic layers thick, that behave differently to their component structures. Or you might directly mimic something biological like a leaf or a bone. Or in my lab we work on opals to have interconnected, active systems. And once you’ve got that coupling of nanostructure and chemistry into a new material, you can start to think about different applications.

At a simple level, you can think about low power devices. So having self-powered health monitoring, just from the ambient temperature of your body. Monitoring in remote hazardous environments where you don’t want to send people in. New systems that will allow us better connectivity and better information gathering.

Our transport systems will use less fuel, and that means less emissions. And We can start to think about connecting our systems so that the heat from the engine is then recaptured and used to power say, the air conditioning units in urban transport networks.

In refrigeration—this is a huge topic—you could just swap out conventional devices that have not really changed for about a hundred years with magnetically-powered technology. They’ll be 50% more efficient. Straight away, 50% efficiency improvement. That, on a grid-scale electricity amount is 3% supply, just from your domestic housing. Twelve megatons of carbon dioxide would be saved with that technology.

But just because we’re thinking about nanoscale materials doesn’t mean we shouldn’t think even bigger. Why don’t we join up all these systems, have thermal recapture with local renewables, local energy storage, to have a completely off-grid factory or data center. We would reduce resource consumption, reduce production costs, reduce environmental impact.

So we’re already seven billion and growing. The demographic’s shifting. More and more of us are moving to cities. And those urban environments are literally hotspots. What if we could think a little bit more cleverly about heat capture. What if we could start to harness some of this energy that’s just being wasted?
So, we’re facing a triple challenge in energy: security, sustainability, and equity. Heat is a huge part of our energy ecosystem. If we even start to make small changes, we will have massive impacts on our energy economy. In the future, these smart nanocomposites will be embedded into everyday devices. They will change the way we think about industrial and urban design.

But right now, it’s still a wasted resource. The tin can you just saw, none of you will be throwing that away. You see that as a resource. What can we do to make people see heat and heat capture as a resource, and make heat capture the new recycling?
Further Reference
Faculty page at Imperial College London
